AKEEGA® offers a well-characterized, consistent, and manageable safety profile1
ADVERSE REACTIONS
Consistent safety established in 2 phase 3, double-blind, placebo-controlled clinical trials comparing AKEEGA® vs standard of care (SoC*)1
In patients with BRCA2 mutations:
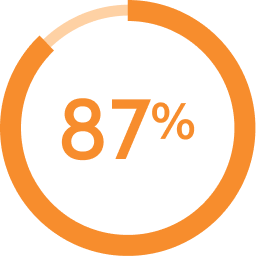 of patients remained on AKEEGA® without discontinuing due to adverse reactions1
of patients remained on AKEEGA® without discontinuing due to adverse reactions1 of patients did not need a dose reduction with AKEEGA®1
of patients did not need a dose reduction with AKEEGA®1Adverse reactions >20% in AKEEGA®–treated patients with a difference of ≥5% vs SoC* (BRCA2 m subgroup)1
| AKEEGA® + P (n=162) | Placebo + AAP (n=161) | |||
|---|---|---|---|---|
| Adverse Reaction | All Grades % | Grade 3-4 % | All Grades % | Grade 3-4 % |
| Hypertensiona | 51 | 31 | 36 | 19 |
| Musculoskeletal paina | 45 | 6 | 58 | 4.3 |
| Constipation | 41 | 0 | 17 | 0.6 |
| Nausea | 30 | 0 | 17 | 0 |
| Fatiguea | 39 | 4.3 | 29 | 3.1 |
| Respiratory tract infectiona | 23 | 0.6 | 13 | 0.6 |
| Arrhythmiaa | 23 | 3.7 | 9 | 2.5 |
aIncludes multiple similar terms.
*SoC = abiraterone acetate + prednisone.
Select laboratory abnormalities >20% that worsened from baseline in patients
who received AKEEGA® (BRCA2 m subgroup)1
| AKEEGA® + P (n=162)a | Placebo + AAP (n=161)a | |||
|---|---|---|---|---|
| Laboratory Abnormality | All Grades % | Grade 3-4 % | All Grades % | Grade 3-4 % |
| Decreased hemoglobin | 74 | 29 | 53 | 1.9 |
| Decreased lymphocyte count | 59 | 20 | 37 | 13 |
| Decreased neutrophil count | 49 | 10 | 19 | 3.1 |
| Decreased platelet count | 41 | 4.9 | 23 | 0.6 |
| Decreased potassium | 38 | 9 | 29 | 10 |
| Increased creatinine | 30 | 1.3 | 16 | 2.5 |
| Increased ALP | 28 | 0.6 | 24 | 3.1 |
| Increased ASP | 24 | 1.3 | 33 | 2.5 |
| Increased blood bilirubin | 22 | 0 | 11 | 0 |
Reference:
- AKEEGA® [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc.
